 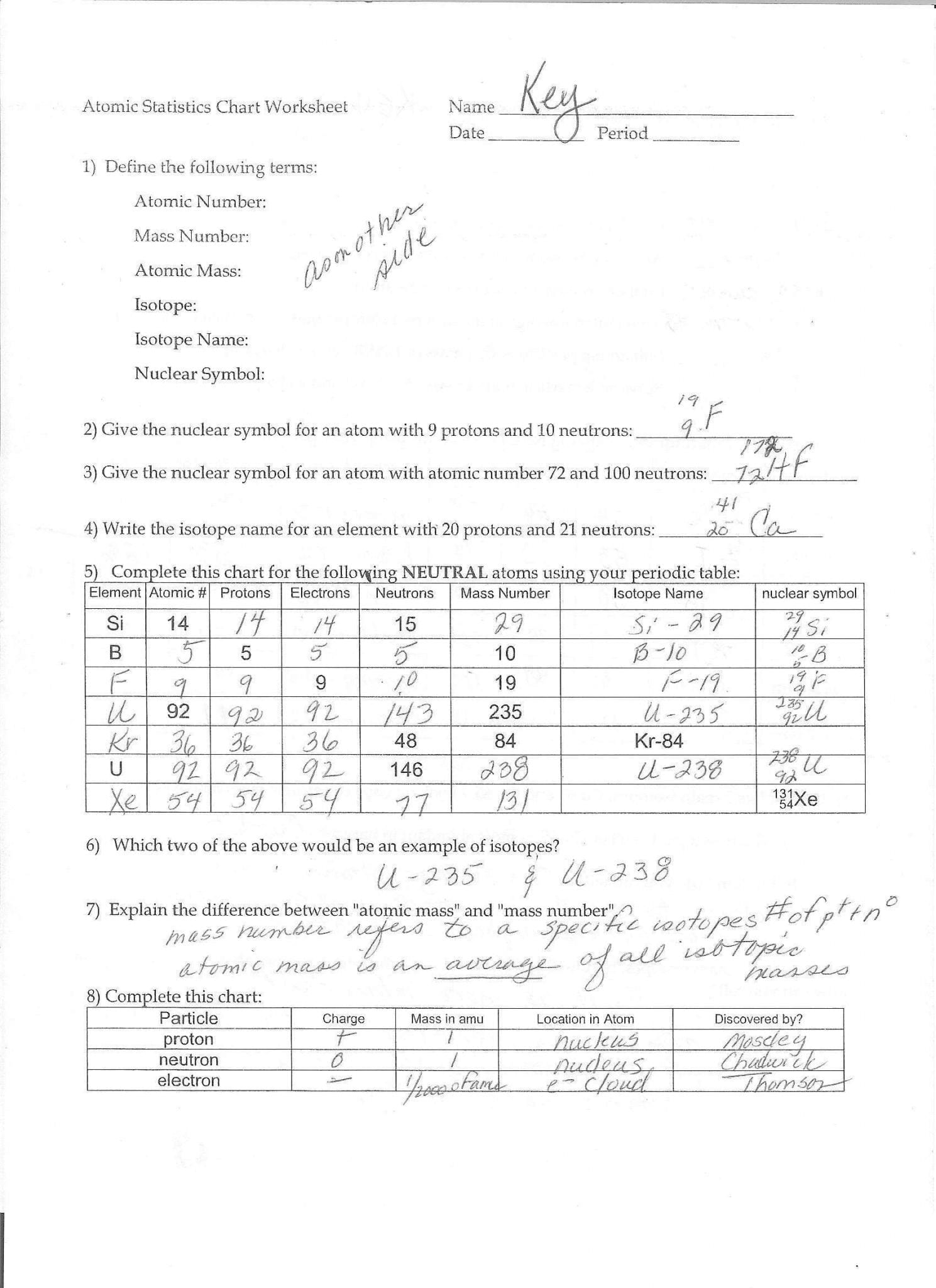 
Has a negative charge and we need to figure out Really what ion we're dealing with because this Given some information about what isotope and Than protons which gives it this negative charge. Plus the neutrons are 32, and it has two more electrons Of sulfur that has a mass number of 32, the protons 
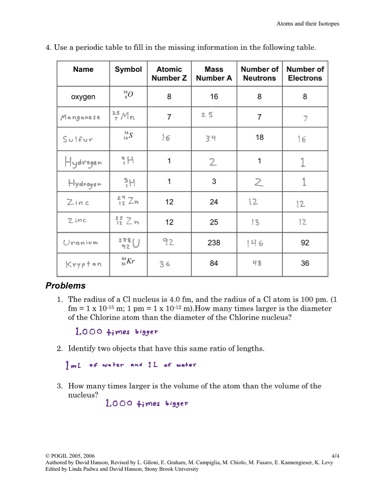
More, we're going to have a negative two charge and Negative charged particles we, and we have two Than protons and since we have a surplus of the But in this case, we haveĪ surplus of electrons. Of protons and electrons, then you would have no charge. Well, the protons have a positive charge. The protons plus the neutrons together, you're going So, this case we haveġ6 protons and we have 16 neutrons, so if you add So, the sulfurs that haveĭifferent number of neutrons, those would be different isotopes. They might have different numbers of neutrons. Sulfur atoms are going to have 16 protons, but Might want to think about is the mass number of Protons by definition is going to be sulfur right over here. Number, what has 16 protons, well anything that has 16 Protons, well we can go right over here to the atomic The number of protons, you should be able to look atĪ periodic table and figure out what element they are talking about. So, an element is defined by the number of protons it has. Pause the video and see if you can figure it outĪnd I'll give you a hint, you might want to use What is the identity of the isotope? And I encourage you to The charge is a positive number when we have more protons than electrons, a negative number when we have more electrons than protons, and 0 when the protons equal the number of electrons.Ĭontains 16 protons, 18 electrons, and 16 neutrons. So the formula would be: C = Z - e, where C is the charge, Z is protons, and e is electrons. The neutrons have a neutral charge so they have no influence on the charge of the atom/ion. The protons of an atom are responsible for its positive charges and the electrons are responsible for the negative charges. Ions are any atoms which has an overall charge which isn't neutral (0). We can use the formula to know them mass number too if we know the number of protons and neutrons by simply adding them together. So, 3 = 1 + N, N = 2, it has two neutrons. For example, If we want to know how many neutrons H-3 (read as hydrogen three or H three) has we know the mass number is 3 and the number of protons is 1 because it's hydrogen and that's its atomic number. So since we have three variables, if we know two we can find the remaining third. So the general formula is: A = Z + N, where A is the mass number, Z is the number of protons, and N is the number of neutrons. The mass number is either written as a superscript in the upper left of the element symbol or hyphenated next to the symbol. Isotopes are usually labeled according to their mass number which is the sum of their protons and neutrons. Isotopes would be atoms of the same element (so the same number of protons), but different numbers of neutrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
